Press Release
When Pain Points in Cross-Border Payment Brings Payment Changes, How Can Hypercard Lead the Trend
Throughout the evolving history of global cross-border payment, cross-border payment is rising with the continuous development of the international division of labor and international exchanges. In the early days, people use precious metals for cross-border payment and clearing, then followed by paper money, and today’s modern electronic transfer and clearing. Cross-border payment is developing gradually towards a rapid, safe and economical trend as the entire international community is engaging in the activities more frequently and science and technology are changing and progressing.
The change of cross-border payment
According to the data, the total amount of global cross-border payment reached $125 trillion in 2018 and is expected to reach $218 trillion in 2022, promising huge profits.

In the existing transfer and remittance system, the transaction is slow and the cost is high with much margin for error; institutions have to coordinate the value transfer between different internal databases, which makes it extremely difficult to settle transactions quickly. This process not only slows down the transaction progress but also requires large working capital, which has a negative impact on the balance sheet of the institution.
As cryptoassets are gradually accepted by traditional finance, digital currency payment is also implementing and applying quickly. The competition around digital currency has just begun across the globe. In 2019, the emergence of Libra has triggered the catfish effect, and legal currency is discussed more enthusiastically all over the world. Countries have taken precautions and speeded up the research on sovereign digital currency. Even the European Central Bank, which did not seem interested before, recently began to discuss the necessity of developing a unified digital currency. According to a report released by the International Monetary Fund in July of the same year, nearly 70% of the world’s central banks are studying sovereign digital currency.
Some fear that Libra may become a strong currency once in circulation. It can be exchanged with the currencies of countries and erodes the fiat currency. If the weak countries make mistakes in regulation, hyperinflation or even de-monetization will likely happen. In the past, a typical example is Zimbabwe who abolished its local currency and was forced to use the US dollar and other currencies.
Traditional payment giants are fostering digital currency payment
Bitcoin was born to destroy the existing monetary system, which many people think is too expensive and exclusive. Given this, it has a much broader value proposition than a deflationary policy and a hard cap of 21 million coins. The new application of blockchain technology also allows anyone to remit money to counterparties around the world in minutes at a low cost.
This function makes bitcoin directly target the existing payment platforms (such as credit card networks and inter-bank messaging systems). While some companies shrug off these concerns, others see the potential and are looking for ways to create value for partners and shareholders.
According to news on February 20, Visa, an international payment giant, has cooperated with 35 leading digital currency platforms or digital wallets.
These institutions are digital currency platforms licensed by the state or regulated by relevant departments, such as the digital payment platform WireX, the digital currency trading platform Coinbase and Fold, cryptoasset lending platform BlockFi, Austria encryption trading platform Bitpanda, Encrypted debit card platform Crypto.com, etc.
Industry insiders said that the cooperation between Visa and digital currency service providers enables consumers to exchange digital currency more quickly and easily. Users can also deposit this money into their Visa certificates in real-time.
When asked why Visa chose the cryptoasset payment, Visa’s executives clearly expressed their optimism about the payment method in his talks with Forbes: “we saw significant innovation in new financial services for consumers holding digital currency. One example is the growth in demand for digital money lending. We are delighted to work with fintech companies like Cred. The company develops new products in this ecosystem and finds new ways for Visa to improve the entrance of fiat currency associated with these products. “
At present, in addition to Visa, MasterCard, Paypal and other international payment tycoons are also fostering digital currency.
Recently, MasterCard stated that it has cooperated with the Central Bank of The Bahamas to launch the world’s first Bahamas prepaid card. The prepaid card allows people to immediately exchange digital currency into traditional Bahamas dollars and pay for goods and services anywhere MasterCard supports. PayPal also claimed to provide cryptocurrency services to the UK market in the coming months.
Cryptoasset service providers speed up the participation in payment
Not only the traditional payment giants are paying attention to cryptoassets payment, but also the asset service providers in the encryption industry are exploring the possibility of payment. HyperBC, a well-known encrypted asset service provider, has launched a comprehensive consumer card HyperCard. After being deposited with digital currency, the card is available in more than 176 countries and more than 50 million merchants worldwide.
As a global standard credit card, HyperCard supports the binding consumption with third-party payment companies by users
Every payment made by HyperCard is secure and consumer privacy is protected by law. HyperCard can transfer money beyond the geographical limit in a second at a low commission, yet with 24/7 service. It is traceable with clear information of all parties. No matter which city you are in, you can use it at all merchants accepting Visa, Master and UnionPay.

In fact, in addition to payment, the most intuitive appealing of digital currency credit cards is it makes encrypted assets purchasing easy and cash out of cryptoassets. In this context, digital currency payment is still a very new track, and the choice of such products is still limited. The main problems are as follows:
1. Only single-currency payment is supported, such as bitcoin
2. Only available in a small number of areas
3. Users have to buy cryptocurrency issued by the card providers before paying
4. Charge a certain percentage of the annual fee
HyperBC also takes this situation into consideration. It is convenient to apply for HyperCard. The digital currency, deposited into HyperCard, can be exchanged into fiat currency in real-time, eliminating the tedious process and the trouble of cash payment, and significantly improving the user-friendliness of digital currency. HyperCard does not charge for KYC verification and only charges a very low commission for each deposit.

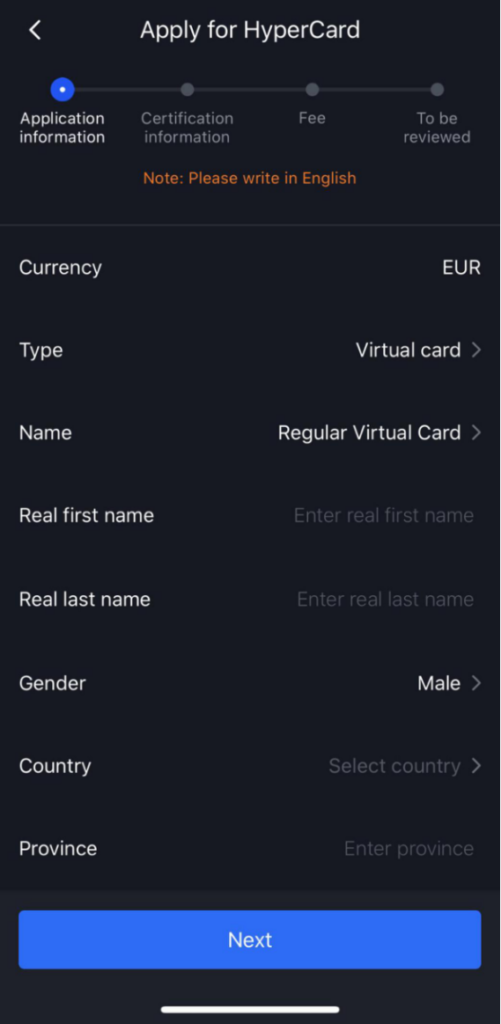
How to apply for HyperCard?
a Download the HyperPay App(https://www.hyperpay.tech/app_down) and register
b Apply for HyperCard
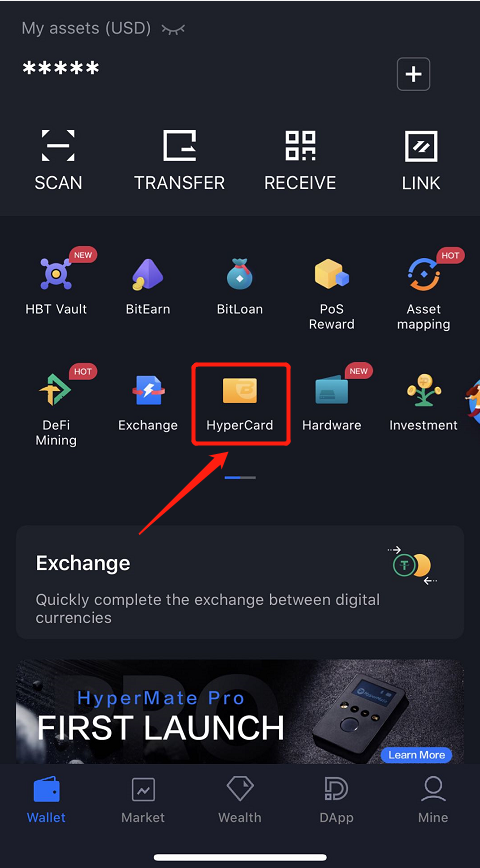
c Submit KYC documents and pass the certification
d HyperCard received
Conclusion
With the rapid development of digital currency and the increasing global acceptance of digital currency, the boundary between fiat currency and digital currency will become narrower. At the same time, digital currency credit card reduces the threshold for traditional users to access digital currency. The selective digital currency assets also avoid their risk in holding digital currency to a certain extent, Whether for investment, quick cash-out, or regular consumption, HyperCard, as a mature digital currency credit card, can enable cardholders to enjoy more convenient services.
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
DRSA – Light It Up Advances Marine Lighting Innovation with High-Performance Dock and Waterfront Solutions
United States, 9th Jun 2026 — DRSA – Light It Up, a trusted marine lighting manufacturer and distributor serving the United States since 1988, continues to lead the industry with advanced, durable, and energy-efficient lighting systems designed specifically for demanding coastal environments.
With over three decades of expertise in marine engineering, DRSA specializes in high-performance LED marine lighting systems engineered for longevity, safety, and aesthetic enhancement across residential docks, marinas, yachts, and commercial waterfront properties. The company’s solutions are built to withstand saltwater exposure, corrosion, UV damage, and extreme weather conditions.
Engineering Excellence in Marine Dock Lighting
DRSA’s core product range includes advanced marine dock lighting systems designed to enhance visibility, safety, and nighttime navigation across waterfront properties. These systems are widely used in marinas, private docks, and coastal developments where reliable illumination is essential.
The company’s lighting solutions include marine LED deck lights and low voltage dock lights that deliver consistent brightness while minimizing energy consumption. Built with marine-grade materials, these fixtures provide long-term durability in harsh saltwater environments.
Premium Piling Cap Lights for Dock Safety and Style
A flagship innovation from DRSA is its premium piling cap lights, engineered to improve both safety and aesthetics across waterfront installations. These lights enhance dock visibility while protecting structural pilings from water damage and environmental wear.
Available in multiple finishes and LED configurations, DRSA’s piling lights support both functional and decorative applications, offering homeowners and marina operators flexible design options that complement modern waterfront architecture.
Advanced Underwater and Boat Lighting Systems
DRSA also delivers cutting-edge boat underwater lighting solutions designed to enhance marine environments with brilliant illumination beneath the waterline. These systems improve nighttime visibility, attract marine life, and create striking visual effects for yachts and recreational boats.
Engineered for durability, these lighting systems are part of DRSA’s broader commitment to innovation in underwater and surface marine illumination.
Specialized Engine Room and Utility Lighting
For internal marine applications, DRSA provides robust marine engine room lights and marine utility lights designed for yachts, commercial vessels, and industrial marine environments.
These systems include advanced LED marine engine room lights and boat engine room lights that ensure high visibility in critical mechanical areas while maintaining energy efficiency and heat resistance.
Innovation in Yacht and Waterfront Lighting Design
DRSA continues to expand its expertise in luxury marine lighting, including specialized yacht engine room lights and integrated lighting systems used in high-end marine vessels.
The company’s technology-driven approach ensures every product meets strict performance standards required for marine safety, operational efficiency, and long-term durability in extreme environments.
Marine Lighting Built for Harsh Coastal Conditions
Every DRSA product is designed for extreme environmental resistance, making it a trusted provider of marine lighting West Palm Beach solutions and other coastal installations across the United States.
Whether installed on private docks or large marina systems, DRSA lighting products are engineered to resist corrosion, UV exposure, saltwater intrusion, and continuous outdoor use. This makes them ideal for both residential waterfront properties and commercial marine infrastructure.
Expanding Applications Across Marine Environments
DRSA lighting systems are widely used across multiple marine environments, including:
- Residential waterfront docks
- Commercial marina developments
- Yacht and boating facilities
- Industrial marine infrastructure
- Underwater dock lighting installations
- Coastal hospitality and resort properties
These applications demonstrate the versatility of DRSA’s engineering approach, ensuring reliable illumination across diverse marine environments.
Commitment to Innovation and Industry Leadership
As a recognized leader in marine illumination, DRSA continues to collaborate with top industry brands such as Lumitec marine lighting to enhance its product ecosystem and expand innovation in LED-based marine systems.
The company remains focused on developing energy-efficient lighting technologies that reduce maintenance costs while improving safety and visual appeal for waterfront properties.
About DRSA – Light It Up
DRSA – Light It Up has been a leading provider of marine lighting solutions since 1988, specializing in dock, deck, engine room, and underwater lighting systems. With a commitment to durability, innovation, and customer satisfaction, DRSA serves residential, commercial, and industrial marine markets across the United States.
Media Contact
Bobby Stone
Business Owner
DRSA – Light It Up
Email: sales@drsa.com
Website: https://www.drsa.com/
Media Contact
Organization: DRSA – Light it up
Contact Person: Bobby Stone
Website: https://www.drsa.com/
Email: Send Email
Address:420 US Highway 1, Suite 8 North Palm Beach, FL 33408
Country:United States
Release id:45861
The post DRSA – Light It Up Advances Marine Lighting Innovation with High-Performance Dock and Waterfront Solutions appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
CellDe Launches Advanced IMEI Verification and Blacklist Detection Solution for the Global Mobile Device Industry
Trusted API empowers retailers, refurbishers, and trade-in providers with real-time lost, stolen, and blacklist detection capabilities
United States, 9th Jun 2026 – CellDe Innovation Labs Pvt. Ltd., a leading provider of mobile device intelligence and trade-in solutions, has announced the expansion of its powerful Device Verification API, designed to help businesses instantly perform IMEI verification and blacklist detection with speed, accuracy, and security.
As the global market for used smartphones and mobile device trade-ins continues to grow, businesses face increasing risks associated with stolen devices, blacklisted IMEIs, activation locks, and inaccurate device information. CellDe’s latest verification solution addresses these challenges by offering a streamlined, API-first platform that enables companies to confidently process devices using only the IMEI number.
The platform delivers real-time insights into device legitimacy and status, helping organizations reduce fraud, improve operational efficiency, and protect customer trust throughout the device lifecycle.
“Our goal is to make device verification smarter, faster, and more accessible for businesses of all sizes,” said Ankur Thakur, CEO of CellDe Innovation Labs Pvt. Ltd. “With the growing demand for B2B mobile device trade-in programs and refurbished smartphones, companies need reliable IMEI verification tools that provide accurate data instantly. CellDe delivers that capability through a secure and highly scalable infrastructure.”
The Device Verification API allows businesses to instantly perform blacklisted IMEI searches and identify whether a device has been reported as lost, stolen, or blocked. The system also provides detailed make and model identification, including brand, storage capacity, and technical specifications, helping organizations accurately classify devices during intake and resale processes.
In addition, the solution supports FMIP (Find My iPhone) lock checks for Apple devices, enabling businesses to determine whether Activation Lock is enabled before accepting or reselling a device. This feature significantly reduces operational complications and financial risks related to locked devices.
CellDe’s verification engine is built for industries that handle high volumes of mobile devices, including:
- Mobile retailers and resellers
- Refurbishers and trade-in providers
- Warehousing and logistics partners
- Repair centers and kiosks
- Buyback platforms and device aggregators
- Insurance and warranty providers
The company states that its API-driven approach allows seamless integration into existing mobile device management systems and trade-in platforms. With detailed documentation, sandbox access, and dedicated technical support, businesses can rapidly deploy the solution without disrupting existing workflows.
One of the key differentiators of CellDe’s IMEI blacklist lookup service is its combination of affordability and enterprise-grade performance. The platform provides lightning-fast results in real time while maintaining pricing that is significantly lower than many competing providers in the market.
Security also remains a major focus for the company. CellDe’s infrastructure is backed by ISO 27001-certified standards, ensuring that all device verification requests and customer data are processed through secure and compliant systems.
The company believes the solution is especially valuable in today’s rapidly expanding secondary smartphone market, where counterfeit devices, fraud, and blacklisted phones continue to create operational and financial risks for businesses worldwide.
By enabling instant lost and stolen phone checks, accurate device identification, and scalable IMEI verification, CellDe aims to simplify the device intake process while helping businesses improve customer confidence and profitability.
The Device Verification API also supports organizations seeking to scale B2B mobile device trade-in operations efficiently. Whether handling hundreds or thousands of IMEI checks daily, the platform is engineered to support high-volume processing with reliability and speed.
CellDe is currently offering businesses the opportunity to explore its API capabilities through trial access and live demonstrations.
For more information or to schedule a demo, visit CellDe Device Verification API.
About CellDe Innovation Labs Pvt. Ltd.
CellDe is a technology company specializing in mobile device intelligence, trade-in automation, and device lifecycle management solutions. The company provides advanced tools and APIs for IMEI verification, blacklist detection, diagnostics, and mobile device management, helping businesses across the global mobile ecosystem streamline operations, reduce fraud, and scale device processing efficiently. Backed by secure ISO-certified infrastructure, CellDe serves retailers, refurbishers, logistics providers, and enterprise partners worldwide.
Media Contact
Organization: CellDe Innovation Labs Pvt Ltd
Contact Person: Ankur Thakur
Website: https://www.cellde.com
Email: Send Email
Country:United States
Release id:45860
The post CellDe Launches Advanced IMEI Verification and Blacklist Detection Solution for the Global Mobile Device Industry appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
CA Detailing Strengthens Position as a Leading UK Destination for Premium Car Detailing Products
United Kingdom, 9th Jun 2026 — CA Detailing, a UK-based specialist retailer of premium automotive detailing products, is reinforcing its position as a go-to destination for enthusiasts, professional detailers, and bodyshops seeking high-performance car care solutions. Operating from its Hitchin warehouse, the company continues to expand its reach nationwide with fast dispatch, expert guidance, and access to globally respected detailing brands.
As demand grows across the UK for premium car detailing products, ceramic coating systems, and professional car care products, CA Detailing is uniquely positioned to serve both trade and enthusiast markets with carefully curated inventory and competitive pricing. The company is an authorized stockist of leading global manufacturers, including Bilt Hamber, Koch Chemie, Gyeon, and CarPro, alongside its own CA Detailing product line developed for performance and value.
A Specialist Alternative to Generalist Retailers
Unlike large generalist retailers, CA Detailing focuses exclusively on the detailing sector, offering deep product knowledge and carefully selected ranges that meet the needs of professional users. This specialist approach allows customers to confidently source everything from car shampoo solutions and advanced surface prep chemicals to high-end microfiber towels and ceramic protection systems.
The company’s team brings hands-on detailing experience, helping customers choose the right products for specific paint systems, correction stages, and protection goals. This level of expertise has positioned CA Detailing as a trusted resource for both beginners exploring the best car cleaning products and professionals scaling their detailing operations.
Authorised Stockist Advantage
A key differentiator for CA Detailing is its status as an authorized UK stockist for several premium international brands. This ensures authenticity, manufacturer-backed quality assurance, and access to the latest product innovations. Many of these professional-grade products can be difficult to source reliably in the UK market, making CA Detailing a preferred supplier for serious detailers and bodyshops.
Popular collections include:
- Koch Chemie Professional Range
- Gyeon Ceramic Coatings & Detailing
- Bilt Hamber Automotive Care Products
Each range is curated to support high-performance detailing workflows, from decontamination and polishing to long-term ceramic protection.
Fast Fulfilment and Trade-Focused Service
CA Detailing’s Hitchin-based warehouse enables rapid nationwide fulfilment, with next-day dispatch available on a large portion of its catalogue. This logistics capability is particularly valuable for trade customers who rely on a consistent supply of car detailing supplies to maintain workshop productivity.
In addition, the company’s pricing structure is designed to remain competitive without compromising product authenticity or service quality. This balance of value and reliability has contributed to growing loyalty among both independent detailers and professional bodyshops.
Strengthening Presence in the Detailing Community
The brand continues to expand its presence across automotive and detailing-focused media, targeting enthusiasts and trade buyers actively researching solutions such as ceramic coating, professional car care products, and specialist detailing chemicals. By aligning content and distribution with high-intent search behaviour, CA Detailing aims to strengthen its authority in the UK automotive care market.
The company also invests in educational content and product guidance, helping customers better understand application techniques, product compatibility, and long-term vehicle protection strategies.
About CA Detailing
CA Detailing is a UK-based premium automotive detailing retailer supplying enthusiasts, professionals, and bodyshops with high-quality car care products. From its Hitchin warehouse, the company offers fast nationwide delivery, expert product advice, and a curated range of professional-grade detailing solutions. CA Detailing is committed to becoming the UK’s most trusted destination for advanced car care and surface protection systems.
Visit the company homepage here: CA Detailing Official Website
Explore the full range of products and collections:
CA Detailing Homepage
For more information, visit https://www.cadetailing.co.uk/ or browse specialist collections for Koch Chemie, Gyeon, and Bilt Hamber products.
Media Contact:
CA Detailing Team
Hitchin, United Kingdom
https://www.cadetailing.co.uk/
Media Contact
Organization: CA Detailing
Contact Person: CA Detailing Team
Website: https://www.cadetailing.co.uk/
Email: Send Email
Contact Number: +442071238995
Country:United Kingdom
Release id:45859
The post CA Detailing Strengthens Position as a Leading UK Destination for Premium Car Detailing Products appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
-
Press Release4 days ago
Post Oak Group Expands Global Investor Network, Deepening Ties with Family Offices and Venture Capital Firms Worldwide
-
Press Release1 day ago
Miles Weber Discusses International Basketball Journey and Continued Growth of Fully Feared Sports
-
Press Release1 week ago
Wijaya Dharmawan Ph.D. Advances Cooperation with NVIDIA Team to Accelerate AI Computing Power and Industrial Applications
-
Press Release4 days ago
BILLSAS Unveils Institutional-Grade GPU Deep Quantitative Models to Enhance Global Market Execution
-
Press Release1 week ago
DEWEL Flea and Tick Prevention for Dogs Brings Prevention-First Care to Fleas Ticks and Mosquitoes
-
Press Release3 days ago
Your Building Team Establishes Itself as Canada’s Go-To Source for Pre-Engineered Steel Building Kits
-
Press Release6 days ago
Author Candace Nicole Releases Gripping New Psychological Thriller Tangled Roots Book One of The Root Series
-
Press Release3 days ago
Ramsey Elkholy on Creating Music That Feels Like a Journey Rather Than a Playlist



