Press Release
Neuralia TMS Broadens Access to Non-Invasive TMS Treatment for Depression
Australia, 27th Jan 2026 – Neuralia TMS, a respected provider of non-invasive neuromodulation therapies, has announced an expansion of access to Transcranial Magnetic Stimulation treatment for individuals living with depression, reflecting continued developments in evidence-based mental health care and increasing demand for non-pharmacological treatment options. The expanded availability is intended to support patients seeking additional clinical pathways under medical supervision.

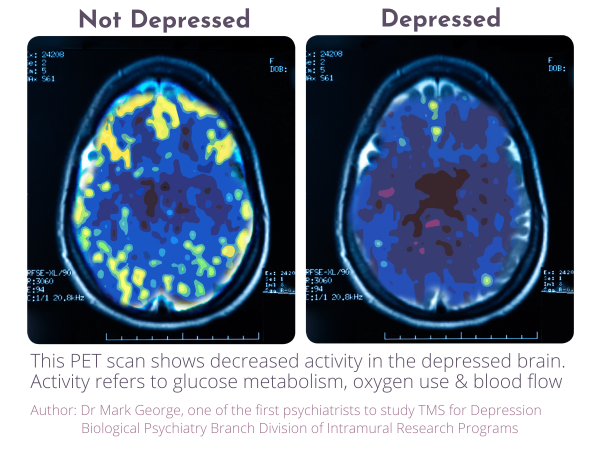
Transcranial Magnetic Stimulation, commonly known as TMS, is a non-invasive procedure that uses targeted magnetic pulses to stimulate specific areas of the brain associated with mood regulation. The treatment does not involve medication or surgery and is delivered in an outpatient setting. Over the past two decades, TMS has been studied extensively and incorporated into treatment guidelines in several countries for individuals with major depressive disorder, particularly where symptoms have persisted despite standard treatment approaches.
The expanded access initiative follows increased clinical interest in treatment pathways that complement existing mental health care models. Depression remains a significant public health concern, with many individuals experiencing ongoing symptoms that affect daily functioning, work participation, and quality of life. In response, mental health services continue to explore a broader range of evidence-based interventions that can be delivered safely and effectively under medical supervision.
Dr Shanek Wick, Medical Director at Neuralia TMS, said the expansion reflects a focus on improving availability of established neuromodulation therapies for appropriate patients. “Transcranial Magnetic Stimulation is a non-invasive treatment option that has been supported by a growing body of clinical research for the management of depression,” Dr Wick said. “Broadening access allows more individuals to be assessed for suitability within a structured medical framework.”
Neuralia TMS provides TMS therapy as part of a comprehensive clinical assessment process, ensuring that treatment plans are informed by individual health history and current clinical needs. Sessions are typically delivered over a course of scheduled appointments, allowing patients to continue with usual daily activities during treatment. Reported side effects are generally mild and temporary, most commonly including scalp discomfort or headache during early sessions.

In addition to depression-focused care, Neuralia TMS offers neuromodulation services for a range of neurological and psychiatric conditions, including anxiety disorders, post-traumatic stress disorder, obsessive-compulsive disorder, chronic pain, and Parkinson’s disease. The clinic also provides transcranial Direct Current Stimulation, an at-home neuromodulation therapy delivered under clinical guidance, and pharmacogenetic testing to support personalised treatment planning based on genetic factors. These services are integrated into broader care considerations rather than positioned as replacements for established mental health treatments.
The expansion of TMS access aligns with wider trends in mental health care that emphasise individualised treatment planning and multidisciplinary approaches. Clinicians increasingly recognise that effective management of depression often involves combining multiple evidence-based strategies, including psychological therapies, medication, and non-invasive interventions. Neuromodulation therapies are being considered as part of this broader continuum of care.
Health sector observers note that non-invasive brain stimulation continues to attract attention as research into brain function and mental health advances. Ongoing clinical studies are exploring how treatment protocols may be refined and how outcomes can be better predicted across different patient groups. Within this context, careful clinical oversight and adherence to established standards remain central to responsible implementation.
Dr Wick said continued evaluation and clinical development will remain important as the field evolves. “Future directions in neuromodulation are expected to involve more precise targeting, improved individualisation of treatment protocols, and closer integration with other mental health services,” Dr Wick said. “Ongoing research and clinical review will play an important role in guiding how these therapies are applied over time.”
Neuralia TMS stated that all patients undergo a comprehensive medical assessment to determine whether TMS is clinically appropriate. The clinic operates in accordance with established medical and professional standards, with an emphasis on informed decision-making and patient safety.
The announcement comes amid continued public discussion about access to timely and evidence-based mental health care. By expanding access to non-invasive TMS treatment for depression, Neuralia TMS aims to contribute to the availability of additional clinical options for individuals and referring practitioners seeking alternatives within a medically supervised setting.
For further information regarding the expanded access to TMS treatment for depression, media enquiries may be directed to Neuralia TMS. The clinic can be contacted by phone on 08 6230 3996 or via email at info@neuralia.com.au.
Media Contact
Organization: Neuralia TMS
Contact Person: Dr Shanek Wick
Website: https://www.neuraliatms.com.au/
Email: Send Email
Contact Number: +61862303996
Country:Australia
Release id:40609
Disclaimer: This announcement is provided for general informational purposes only and does not constitute medical advice. Transcranial Magnetic Stimulation suitability and outcomes vary by individual and should be determined through assessment by a qualified medical professional.
The post Neuralia TMS Broadens Access to Non-Invasive TMS Treatment for Depression appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
Galatea by Inkitt Celebrates 100+ Five-Star Reviews Amid Rising Popularity
California, USA, 3rd May 2026, ZEX PR WIRE — Galatea, the rapidly growing digital reading platform developed by Inkitt, has surpassed 100 five-star reviews, marking a significant milestone in its continued rise among global readers. The achievement highlights strong user satisfaction and reinforces Galatea’s position as more than just a reading app. As part of Inkitt’s broader story-to-screen ecosystem, Galatea plays a central role in transforming reader-driven content into multi-format entertainment, extending all the way to television through CandyJar.
This milestone arrives at a time when audiences are actively seeking more flexible and engaging ways to consume stories. Galatea has responded by offering a format that aligns with modern habits while still preserving the emotional depth that readers expect from strong storytelling.
A Milestone That Reflects Real Reader Loyalty
Reaching over 100 five-star reviews signals more than short-term success. It reflects consistent reader satisfaction and an ability to maintain engagement over time. Users frequently highlight how seamlessly the platform fits into their daily routines, whether they are reading during short breaks, while commuting, or winding down at night.
This accessibility has helped Galatea build a loyal user base. Readers are not simply downloading the app and leaving. They return regularly to continue stories, explore new releases, and stay connected to ongoing narratives. The growing number of five-star reviews demonstrates that users are finding lasting value in the experience.
In an environment where attention is divided across multiple platforms, this level of retention is notable. Galatea has managed to create a product that readers continue to engage with long after their first interaction.
A Reading Experience Designed for Modern Habits
Galatea has built its platform around the idea that reading should adapt to the reader. Instead of presenting long chapters that require uninterrupted time, the platform delivers stories in episodic segments that are easy to consume. This structure makes reading more approachable and encourages consistent engagement.
The platform also offers multiple formats within a single story. Readers can move seamlessly between ebook and audiobook modes, allowing them to switch based on their situation. In addition, Galatea includes chat-style storytelling, where narratives unfold through text-message-like conversations. This format introduces variety and reflects how people naturally communicate today.
Audio is another defining feature of the platform. Full narration and carefully designed sound elements support the storytelling experience and add depth to each scene. Rather than acting as a background feature, audio plays an active role in shaping how stories are experienced.
Together, these features create a reading environment that feels flexible, engaging, and suited to modern lifestyles.
A Story-to-Screen Pipeline That Sets It Apart
Galatea’s growth is closely tied to Inkitt’s larger story-to-screen model, which differentiates it from traditional reading platforms. The process begins on Inkitt, where writers share their work and reader engagement determines which stories gain traction. The most promising titles are then published on Galatea, where they reach a wider audience in enhanced formats.
From there, the most successful stories move into television development on CandyJar. This structured pipeline allows content to evolve naturally, moving from written storytelling to screen adaptation based on proven audience demand.
This model offers a clear pathway for writers. Instead of navigating uncertain publishing routes, authors can see their work progress step by step, guided by reader response. It also ensures that the stories adapted for television already have a built-in audience, reducing risk and increasing the likelihood of success.
For readers, the pipeline adds another layer of engagement. Stories they discover on Galatea have the potential to expand into visual formats, creating a deeper connection to the content. This continuity strengthens the overall experience and sets the platform apart in a crowded market.
Content That Reflects What Readers Actually Want
Galatea’s content strategy is built on real user behavior. By leveraging data from Inkitt, the platform identifies stories that already resonate with readers. This ensures that the content available on Galatea aligns closely with audience preferences.
Genres such as romance, fantasy, thriller, drama, and Young Adult continue to perform strongly, reflecting consistent demand across a wide range of readers. At the same time, the platform introduces new voices and perspectives, maintaining a balance between familiar themes and fresh ideas.
This approach reduces the uncertainty often associated with content selection. Instead of relying on predictions, Galatea uses measurable engagement to guide its decisions. The result is a library that feels both curated and relevant.
For writers, this system creates meaningful opportunities. Success is determined by reader response, allowing emerging authors to gain visibility and build an audience based on the strength of their work.
Listening to Feedback and Improving the Experience
The increase in five-star reviews also reflects Galatea’s responsiveness to its users. Feedback plays a direct role in shaping the platform’s development. Readers frequently mention features such as customizable settings, smooth navigation, and the ability to switch between formats without interruption.
These elements contribute to a user-friendly experience that appeals to a broad audience. Small improvements, driven by real feedback, continue to enhance the platform’s usability.
Galatea’s ability to adapt has become a key strength. As user expectations evolve, the platform remains focused on refining its features while maintaining the core aspects that readers value.
Meeting the Demand for Flexible Storytelling
Modern readers are looking for flexibility, and Galatea has positioned itself to meet that demand. The platform allows users to engage with stories in different ways, depending on their preferences and schedules.
Whether through short reading sessions, audiobook listening, or interactive chat-based formats, users can choose how they want to experience each story. This adaptability makes it easier to stay engaged without disrupting daily routines.
The ability to move between formats also adds to the overall experience. A reader can begin a story in text form, continue it as an audiobook, and explore chat-style interactions within the same narrative. This level of flexibility keeps the platform dynamic and appealing.
Building a Community Around Stories
As Galatea continues to grow, it is also building a strong and engaged community. The increasing number of five-star reviews reflects not only individual satisfaction but also a shared enthusiasm among users.
Readers actively recommend the platform, share feedback, and contribute to its visibility. This organic growth has played an important role in expanding Galatea’s reach.
For writers, this community creates valuable opportunities for connection and recognition. Authors can engage with readers directly and see how their work resonates in real time. This interaction strengthens the relationship between creators and their audience.
Each positive review adds to the platform’s credibility and encourages new users to explore what it has to offer.
Looking Ahead
Surpassing 100 five-star reviews marks an important milestone, but it also signals continued growth. Galatea remains focused on expanding its content library, improving its features, and strengthening its role within Inkitt’s story-to-screen ecosystem.
The integration with CandyJar creates new possibilities for content development, allowing stories to move beyond the app and reach wider audiences. This connection positions Galatea as a key part of a larger entertainment strategy rather than a standalone platform.
As more readers discover the app, the platform is expected to continue building momentum and attracting new users. The steady increase in positive feedback suggests that it is meeting the expectations of its audience and adapting effectively to changing preferences.
Conclusion
Galatea’s achievement of over 100 five-star reviews reflects its ability to deliver a reading experience that aligns with modern expectations. By combining flexible formats, data-driven content, and a clear path from story to screen through CandyJar, the platform offers something distinct in the digital reading space.
As part of Inkitt’s broader vision, Galatea represents more than a reading app. It is a central piece of a storytelling ecosystem where ideas begin with readers and have the potential to grow into larger forms of entertainment. With strong user engagement and a clear direction for growth, Galatea continues to establish itself as a platform that understands both storytelling and the people who enjoy it.
For more information, please feel free to visit https://galatea.com/
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
Milestone Launches Project : Tsavkisi, The First Design-Code Community Near Tbilisi
Milestone has launched Project : Tsavkisi, a residential land-plot development situated on a hillside in the Tsavkisi area, just an 18-minute drive from Tbilisi, Georgia. The project comprises 63 individual land plots, each offering panoramic views of the surrounding forest and the city.
Tbilisi, Georgia, 2nd May 2026 – Milestone has launched Project : Tsavkisi, a residential land-plot development situated on a hillside in the Tsavkisi area, just an 18-minute drive from Tbilisi, Georgia. The project comprises 63 individual land plots, each offering panoramic views of the surrounding forest and the city.

Project : Tsavkisi is a thoughtfully planned community with a unified architectural identity, seamlessly integrated into the natural landscape. It is designed for those who wish to build a residence in a scenic setting while remaining just minutes away from central Tbilisi.
The presale is now open.
The development spans 10 hectares, with 24,000 m² dedicated to curated shared public spaces, including children’s playgrounds, sports courts, and forest walking paths. The project includes professionally maintained shared areas and green zones, along with 24/7 secured territory.
The architectural vision of Project : Tsavkisi is rooted in a landscape-oriented approach. To create a unique living environment, the project has developed a set of architectural guidelines – a Design Code, where every element is thoughtfully connected to the next. The Code extends from façade materials and roofing guidelines to color selection. A recommended palette of muted, earth-derived tones is designed to maintain visual harmony with the natural landscape. The project adopts a low-density development strategy that maintains visual openness and integrates seamlessly with the natural terrain, avoiding high retaining walls and other intrusive structural elements.
Homeowners are offered the flexibility to develop individual designs in accordance with the Design Code or to select from a catalogue of nine pre-designed residential houses. They have been created in collaboration with established architectural studios, recognized for their experience in residential design and landscape-driven planning.
The project is designed for residents who lead active urban lives but seek a home in a quiet, natural setting, without sacrificing their connection to the city. Project : Tsavkisi sets a benchmark for holistic suburban development, where each residence contributes to a unified architectural identity.
About Milestone
Milestone is a Georgian real estate development and management company based on innovative vision and ideas, defining environments for quality life. Expanding its portfolio, the company is currently developing a new residential project in the heart of Tbilisi, set to be introduced soon.
Media Contact
Organization: Milestone
Contact Person: Lasha Eliadze
Website: https://www.projecttsavkisi.ge/
Email:
info@milestone.ge
Contact Number: +995598399899
City: Tbilisi
Country:Georgia
Release id:44575
The post Milestone Launches Project : Tsavkisi, The First Design-Code Community Near Tbilisi appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
Dark Watch Integration with Leading Service Platform Sets a New Standard of Care in Therapist Safety
Integration embeds real-time safety intelligence into therapists’ booking and payment workflows—helping therapy practices and service-based businesses identify and prevent high-risk interactions before appointments begin.
United States, 2nd May 2026 —May 1, 2026—Dark Watch, the embedded safety intelligence layer for modern commerce, today announced that its integration with a leading service-based platform is now live. The integration introduces a proactive safety capability designed to help prevent assaults and high-risk incidents in therapist-client environments.
Service-based businesses, particularly those in the wellness, spa, and personal care sectors, often involve private, one-on-one interactions between staff and clients. These environments are built on trust, but they can also expose therapists to elevated risk, such as from inappropriate behavior, harassment, boundary violations, and, in some cases, physical assault. Despite this, most booking and payment systems provide little to no visibility into potential risk before an appointment begins.
Historically, businesses have relied on reactive measures to mitigate these risks, e.g., incident reporting, internal notes, or post-event responses. The Dark Watch integration shifts that model upstream, enabling businesses to identify potential risk signals before an appointment is confirmed or a client arrives.
“A service-based business should not have to rely on reactive measures to reduce risk,” said Noel Thomas, CEO of Dark Watch. “Therapists are operating in one-on-one settings where the risk is real, and expectations are changing. Businesses shouldn’t have to choose between experience and protection. Dark Watch embeds security directly into the booking and payment layer, delivering real-time risk mitigation at the moment it matters most. The result is a new standard of care—one that protects therapists, strengthens brand trust, and delivers measurable safety benefits without disrupting the client experience.”
Through the integration, Dark Watch’s intelligence layer is embedded directly into booking and payment workflows. As appointments are scheduled, the system evaluates a range of risk signals that help identify patterns associated with prior inappropriate behavior, unsafe interactions, or indicators that warrant additional review.
The integration enables businesses to receive discreet, real-time insights at the moment of booking—before a therapist is assigned, before a client arrives, and before a potentially unsafe interaction occurs.
For operators, this creates a new layer of protection: improving therapist safety by identifying high-risk appointments early, increasing operational confidence when interacting with new clients, reducing the likelihood of incidents, and supporting staff retention in roles where safety concerns can drive turnover.
By embedding this capability directly into the platform layer, businesses can adopt proactive safety without adding friction to the booking experience or requiring additional tools. The system operates quietly in the background, preserving the client experience while enhancing staff protection.
The launch reflects a broader shift across the experience economy. Platforms are increasingly expected to safeguard not only transactions, but also the people behind them. As awareness grows around therapist safety in one-on-one service environments, proactive protection is becoming a critical expectation and an emerging standard of care.
Dark Watch’s technology is gaining traction across a growing set of platforms spanning payments, financial services, and real-world commerce systems. With multiple platform deployments now live or underway, the company continues to expand its footprint as a safety intelligence layer embedded directly into the infrastructure that powers everyday interactions.
Together, these deployments are establishing a new standard: protecting therapists and staff before the interaction begins.
For more information, visit https://darkwatch.io
About Dark Watch
Dark Watch delivers an embedded safety intelligence layer for payment platforms, financial institutions, and real-world commerce systems. The platform leverages advanced AI and proprietary intelligence sources to identify risk signals tied to unsafe behavior, harassment patterns, and high-risk interactions—before transactions occur.
Media Contact
Organization: Dark Watch
Contact Person: Noel Thomas
Website: https://darkwatch.io
Email: Send Email
Country:United States
Release id:44597
The post Dark Watch Integration with Leading Service Platform Sets a New Standard of Care in Therapist Safety appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
-
Press Release3 days ago
Representatives from More Than 40 Countries Discuss New Models of Global Growth in Moscow
-
Press Release1 week ago
AntNest $1,000,000 Genesis Airdrop is LIVE! Play games to Win Big!
-
Press Release6 days ago
Singapore’s Lucky Gemstones Launches Personalized Custom Gemstone Ring Service, Letting Every Love Story Bloom in an Enchanted Forest
-
Press Release6 days ago
TianYi High-Tech Launches TA Series Carbide Roller Rings for High-Speed Rolling Mills
-
Press Release4 days ago
Anyang Wire Mesh Launches New Epoxy and Oil-Water Separation Mesh for Global Filtration
-
Press Release4 days ago
Excimerlight Breakthrough UV Technology: 172nm EUV 222nm and 254nm Pet Disinfection Lamp
-
Press Release2 days ago
Breescape Cooling Products Earn Prestigious Good Housekeeping Seal
-
Press Release2 days ago
Crypto Office Makes Working with Cryptocurrencies Simple and Secure



