Press Release
CMS (867.HK/8A8.SG): NDA for Additional Indication Atopic Dermatitis (AD) for Ruxolitinib Phosphate Cream Accepted and Granted Priority Review in China
SHENZHEN, CHINA – China Medical System Holdings Limited (“CMS” or the “Group”) is pleased to announce that its subsidiary, Dermavon Holdings Limited (“Dermavon”, an innovative pharmaceutical company specialized in skin health which is applying for a separate listing on the Main Board of The Stock Exchange of Hong Kong Limited, please refer to the announcement published by the Group on 22 April 2025 for details) received the acceptance from the National Medical Products Administration of China (NMPA) for the New Drug Application (NDA) of ruxolitinib phosphate cream (the “Product”) for the treatment of mild to moderate atopic dermatitis on 24 February 2026. The Product is indicated for the topical short-term and non-continuous chronic treatment of mild to moderate atopic dermatitis in non-immunocompromised adult and pediatric patients 2 years of age and older whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. The NDA has been approved for inclusion in the Priority Review List by the Center for Drug Evaluation (CDE) of the NMPA based on its qualification as a “new variety, dosage form and specification of pediatric drug that conforms to the physiological characteristics of children”, which is expected to accelerate the Product’s review process for marketing approval in the AD indication.
Impressive phase III trial data in AD
Its Priority Review is expected to accelerate its NDA approval
Ruxolitinib phosphate cream was approved for marketing by the NMPA in January 2026, becoming the first and only targeted drug approved in China for the treatment of vitiligo. The acceptance of this NDA for the additional indication of AD marks a key milestone in the Product’s expansion into multiple therapeutic areas.
Ruxolitinib phosphate cream has achieved positive results in a randomized, double-blind, placebo-controlled Phase III clinical trial in China for mild to moderate AD.
- Robust Efficacy: The Product successfully met its primary endpoint, demonstrating that a significantly higher proportion of subjects treated with ruxolitinib phosphate cream achieved IGA (Investigator’s Global Assessment) of 0 or 1 with at least two grades of reduction from baseline at week 8, compared with placebo (63.0% vs 9.2%, P < 0.001). For the key secondary endpoint, the proportion of subjects achieving at least a 75% improvement from baseline in the Eczema Area and Severity Index score (EASI 75) of treatment with ruxolitinib phosphate cream was also significantly higher than that of the placebo group, at week 8 (78.0% vs 15.4%, P < 0.001).
- Favorable Safety Profile: The severity of treatment-emergent adverse events (TEAE) during the treatment period was mostly mild or moderate, with no TEAEs leading to discontinuation of the study drug. Overall, ruxolitinib phosphate cream was safe and well-tolerated.
Furthermore, the NDA has been approved for inclusion in the Priority Review List. According to relevant NMPA regulations, the review timeline for marketing applications subject to Priority Review and Approval Procedure is substantially shortened—from the standard 200 days to 130 days. This significant reduction in the review timeline is expected to accelerate the marketing process of ruxolitinib phosphate cream for the AD indication, benefiting a wide range of patients at an earlier date.
Building an integrated AD solution,
strengthening Dermavon’s leadership in dermatology
AD is a chronic, recurrent and inflammatory dermatologic disease, with the main clinical manifestations of dry skin, chronic eczema-like lesions and obvious itching or pruritus, which may seriously affect the quality of life of patients. It is estimated that there were over 54 million AD patients in China as of 2024. Based on SCORAD scores, mild to moderate AD accounts for 98% of these cases, representing over 52.5 million patients[1]. Topical drugs are the most basic treatment for AD. Traditional topical medications such as topical corticosteroids (TCS) and topical calcineurin inhibitors (TCIs) have clinical pain points with long-term adverse reactions or limited efficacy, therefore novel treatments are urgently needed.
Dermavon has developed a comprehensive “treatment + care” integrated solution for AD. In addition to the topical formulation ruxolitinib phosphate cream for mild-to-moderate AD, the portfolio also includes the injectable biological agent Comekibart Injection (MG-K10) for moderate-to-severe AD, the oral small molecule targeted drug CMS-D001 for moderate-to-severe AD, and the Heling Soothing Product Series for daily repair, to meet the management needs of AD patients from treatment to daily care.
Simultaneously, the acceptance of the NDA for the AD indication will strengthen Dermavon’s strategic layout in the field of skin treatments and create synergies with its commercialized innovative drug ILUMETRI (tildrakizumab injection), commercialized exclusive drug Hirudoid (mucopolysaccharide polysulfate cream), and a series of innovative drugs under development and dermatological skin care products, in terms of expert network and market resources, thereby potentially enhancing Dermavon’s market competitiveness and brand influence in the field of skin health.
More Information About Ruxolitinib Phosphate Cream
Ruxolitinib phosphate cream (Opzelura®), a novel cream formulation of the selective JAK1/JAK2 inhibitor ruxolitinib developed by Incyte. In China, ruxolitinib phosphate cream is approved for the treatment of non-segmental vitiligo with facial involvement in children aged 12 years and older and adult patients. In the U.S., ruxolitinib phosphate cream is the first topical JAK inhibitor approved by the U.S. Food and Drug Administration (FDA) for the topical treatment of non-segmental vitiligo in patients aged 12 years and older, and for the topical short-term and non-continuous chronic treatment of mild to moderate atopic dermatitis in non-immunocompromised adult and pediatric patients 2 years of age and older whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable[2]. In Europe, ruxolitinib phosphate cream is approved for the treatment of non-segmental vitiligo with facial involvement in adults and adolescents from 12 years of age[3].
The Group, through its subsidiary of Dermavon entered into a Collaboration and License Agreement with Incyte for ruxolitinib phosphate cream on 2 December 2022, obtaining an exclusive license to develop, register and commercialize the Product in Mainland China, Hong Kong Special Administrative Region, Macau Special Administrative Region, Taiwan Region and eleven Southeast Asian countries (the “Territory”) and a non-exclusive license to manufacture the Product in the Territory. The subsidiary of Dermavon has sublicensed the relevant rights for the Product outside of Mainland China to the Group (excluding Dermavon and its subsidiary).
Incyte has worldwide rights for the development and commercialization of ruxolitinib phosphate cream, marketed in the United States and Europe as Opzelura®. Opzelura® and the Opzelura® logo are registered trademarks of Incyte.
About CMS
CMS is a platform company linking pharmaceutical innovation and commercialization with strong product lifecycle management capability, dedicated to providing competitive products and services to meet unmet medical needs.
CMS focuses on the global first-in-class (FIC) and best-in-class (BIC) innovative products, and efficiently promotes the clinical research, development and commercialization of innovative products, enabling the continuous transformation of scientific research into clinical practices to benefit patients.
CMS deeply engages in several specialty therapeutic fields, and has developed proven commercialization capabilities, extensive networks and expert resources, resulting in leading academic and market positions for its major marketed products. CMS continues to promote the in-depth development in its advantageous specialty fields, strengthening the competitiveness of the cardio-cerebrovascular/ gastroenterology/ ophthalmology/ skin health businesses, bringing economies of scale in specialty fields. Among them, the skin health business (Dermavon) has become a leading enterprise in its field, and is proposed to be listed independently on the SEHK. Meanwhile, CMS continuously promotes the operation and development of its integrated R&D, manufacturing and commercialization chain in Southeast Asia and the Middle East, capturing growth opportunities in emerging markets to support the high-quality and sustainable development of the Group.
Reference:
1. China Insights Consultancy’s industrial report
2. The U.S. FDA approval information can be found on the Incyte official website, as follows:
3. The EMA approval information can be found on the Incyte official website, as follows:
CMS Disclaimer and Forward-Looking Statements
This press release is not intended to promote any products to you and is not for advertising purposes. This press release does not recommend any drugs, medical devices and/or indications. If you want to know more about the diagnosis and treatment of specific diseases, please follow the opinions or guidance of your doctor or other medical and health professionals. Any treatment-related decisions made by healthcare professionals should be based on the patient’s specific circumstances and in accordance with the drug package insert.
This press release which has been prepared by CMS does not constitute any offer or invitation to purchase or subscribe for any securities, and shall not form the basis for or be relied on in connection with any contract or binding commitment whatsoever. This press release has been prepared by CMS based on information and data which it considers reliable, but CMS makes no representation or warranty, express or implied, whatsoever, and no reliance shall be placed on, the truth, accuracy, completeness, fairness and reasonableness of the contents of this press release. Certain matters discussed in this press release may contain statements regarding the Group’s market opportunity and business prospects that are individually and collectively forward-looking statements. Such forward-looking statements are not guarantees of future performance and are subject to known and unknown risks, uncertainties and assumptions that are difficult to predict. Any forward-looking statements and projections made by third parties included in this press release are not adopted by the Group and the Company is not responsible for such third-party statements and projections.
Media Contact
Brand: China Medical System Holdings Ltd.
Contact: CMS Investor Relations
Email: ir@cms.net.cn
Website: https://web.cms.net.cn/en/home/
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
Trakx expands Canton presence as CTIs become available through Five North’s Loop wallet

Trakx, the leading platform for crypto-index trading, announced that its Crypto Tradable Indices (CTIs) are now available through Five North’s Loop wallet on Canton, enabling CTIs to be held and transferred on-chain through a Canton-native wallet environment. The milestone marks a new step in the evolution of Trakx’s index products beyond the Trakx platform and into the broader infrastructure of the Canton ecosystem.
With this integration, Trakx Crypto Tradable Indices gain a new layer of accessibility and portability on-chain. By becoming available through Loop, Five North’s wallet product for Canton, CTIs can now be held and transferred across the Canton Network, an institutional-grade blockchain environment designed for privacy and interoperability.
The development is strategically significant for Trakx as it continues to strengthen the infrastructure and distribution layer surrounding its crypto index products. While CTIs have historically been accessed primarily through the Trakx platform, their availability through Loop reflects a broader shift toward interoperability, partner integration, and professional distribution models across digital assets.
“This milestone is about more than wallet availability,” said Lionel Rebibo, CEO at Trakx. “It reflects the direction we believe digital asset products need to take: beyond standalone platform access and toward deeper integration within institutional-grade blockchain infrastructure. By making CTIs available through Loop on Canton, we are taking a concrete step toward broader B2B and B2B2C distribution, stronger interoperability, and a more scalable framework for partners looking to integrate structured digital asset exposure into their own environments.”
Canton Network plays an important role in that strategy. Designed as a privacy-enabled blockchain network with a strong institutional orientation, Canton provides the type of infrastructure environment that can support on-chain issuance, asset portability, and interoperable financial workflows. For Trakx, this creates a foundation for CTIs to evolve from platform-native products into assets that can be integrated into broader access and distribution models.
Five North‘s role in the Canton ecosystem gives additional strategic relevance to the launch. As a builder and operator of core infrastructure on Canton Network, including wallets, explorers, and validator systems, Five North occupies a meaningful position within the network. Its Loop wallet provides the practical layer through which CTIs can now be held and transferred on-chain, helping translate tokenization into real ecosystem usability.
For Trakx, the move is especially relevant in the context of partner-led adoption. The company sees increasing value in enabling B2B platforms, B2B2C distributors and other professional intermediaries to access more structured and infrastructure-ready models for digital asset exposure. In that sense, the integration is not simply a technical enhancement, but part of a broader distribution strategy focused on making CTIs easier to integrate, distribute, and use across professional environments.
As digital asset markets mature, infrastructure, accessibility, and interoperability are becoming just as important as product design. With CTIs now available through Five North’s Loop wallet on Canton, Trakx is advancing its objective of building not only better crypto index products, but also better infrastructure pathways for those products to be held, transferred and integrated across the next generation of on-chain finance.
About Trakx
Trakx is a crypto index investing platform offering Crypto Tradable Indices designed to provide structured exposure to digital asset markets. Its mission is to make crypto investing simpler, more transparent, and more accessible through index-based products built for both individual and professional use.
Contact Information
Gary Rebibo | CMO at Trakx | gary@trakx.io
Website | LinkedIn | X/Twitter | Discord | Telegram
Media Contact
Organization: Trakx
Contact Person: Gary Rebibo
Website: https://trakx.io/
Email: Send Email
City: Paris
State: France
Country:France
Release id:44145
The post Trakx expands Canton presence as CTIs become available through Five North’s Loop wallet appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
General Compute Launches ASIC-First Inference Cloud for Autonomous AI Agents
General Compute today announced its inference cloud platform built for AI agents, working with early partners now ahead of general availability on May 15, 2026. The platform runs on purpose-built AI accelerators rather than general-purpose GPUs. More information is available at generalcompute.com.
SAN FRANCISCO — April 18, 2026 — General Compute Inc. today announced its inference cloud platform, which is designed for AI agent workloads. The company is working with early partners now, with general availability scheduled for May 15, 2026.
The platform runs on purpose-built AI accelerators rather than general-purpose graphics processors. Its architecture separates the prefill and decode stages of inference processing, allowing each stage to be scaled independently based on workload.
The platform is built to serve AI agents that make high volumes of LLM inference and tool calls, including AI agents that provision their own compute programmatically.
“The last 20 years we built for developers, the next 20 we will build for agents. On General Compute, AI agents can sign up on their own and provision their own inference. Our docs and API are optimized for both human and AI agent consumption,” said Jason Goodison, co-founder and Chief Technology Officer of General Compute.
Platform Overview
The platform offers an industry-standard API, allowing developers to integrate it into existing applications with minimal code changes. AI agents and developers alike can sign up, provision API keys, and begin making inference calls programmatically.
At launch, the platform will offer access to a range of open-source LLMs across multiple model families and parameter sizes. Customers can also deploy their own models on the company’s infrastructure.
Infrastructure
General Compute’s data center infrastructure operates on hydroelectric power. The company states that its accelerator hardware is air-cooled, and that its racks operate at lower power densities than comparable installations built on general-purpose processors.
The company publishes technical performance data for its platform on its website.
Availability
General Compute is working with early partners now, with general availability beginning May 15, 2026. Enterprise inquiries regarding dedicated infrastructure, service level agreements, and capacity planning may be directed to jason@generalcompute.com
About General Compute
General Compute Inc. is an inference cloud company headquartered in California. The company was founded by Jason Goodison and Finn Puklowski.
Contact
Jason Goodison, Co-founder and Chief Technology Officer General Compute Inc. jason@generalcompute.com generalcompute.com
Media Contact
Organization: General Compute Inc
Contact Person: Jason Goodison
Website: https://generalcompute.com
Email:
jason@generalcompute.com
Contact Number: +14257537667
Address:440 North Barranca Avenue
Address 2: 3780
City: Covina
State: California
Country:United States
Release id:44077
The post General Compute Launches ASIC-First Inference Cloud for Autonomous AI Agents appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
Press Release
Tempo Mails Launches Temporary Email Address Platform for Instant Disposable Inbox Access

MIAMI, Florida, United States — April 18, 2026 — Tempo Mails today announced the launch of its free temporary email address platform, designed to help users create disposable email addresses instantly for sign-ups, verification codes, online trials, and privacy-focused browsing. The web-based service provides a temporary mailbox without registration and supports real-time inbox access for users who need a burner email without linking it to a personal account.
Available at tempomails.com, the platform is built for users who want a faster way to manage one-time online interactions while keeping their primary inbox separate from promotional messages, verification traffic, and unwanted follow-up emails. According to the company’s website, the service creates a temporary email address on page load, supports inbox history controls, and allows users to refresh, delete, or add new addresses from the interface.
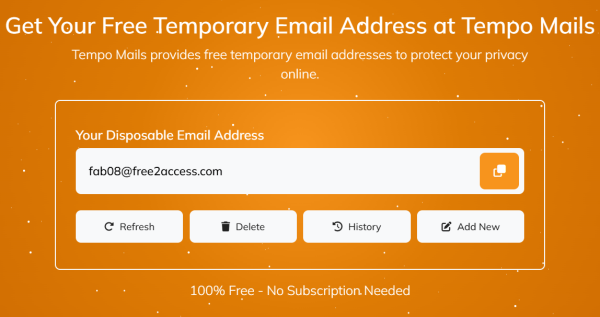
Tempo Mails said the service is intended for common online use cases such as account verification, e-commerce sign-ups, app testing, temporary registrations, and limited anonymous communication. The platform also supports temporary email send and receive workflows where users need short-term inbox access to receive OTPs, confirmation links, and service-related messages without using a permanent account. The site states that no sign-up is required and that inboxes are auto-cleared after use.
The service includes features such as custom email alias options, real-time inbox refresh, auto-delete functionality, and multi-domain address generation. Tempo Mails also states that the platform is free to use and designed to work worldwide, giving users access to a disposable inbox through a browser-based interface rather than a downloadable application.
“With Tempo Mails, the goal is to make privacy-focused email access simple for everyday users,” said James at Tempo Mails. “People often need a temporary mailbox for a single task, such as receiving a code or completing a registration, without exposing their personal inbox to long-term marketing traffic.”
The launch comes as more users look for practical ways to separate account creation and one-time online activity from their primary email accounts. By offering disposable email addresses that can be created in seconds, Tempo Mails is positioning the platform as a lightweight privacy tool for consumers, testers, developers, and online shoppers who need a burner email for short-term use.
About Tempo Mails
Tempo Mails is a web-based temporary email service that provides free temporary email address generation for users who want short-term inbox access without registration. The platform offers disposable email addresses, real-time inbox refresh, custom alias controls, and temporary mailbox features designed for account verification, online trials, and privacy-focused browsing. The service is available through tempomails.com and is designed for fast access across devices.
Media Contact
Organization: Tempo Mails
Contact Person: James
Website: https://tempomails.com/
Email:
contact@tempomails.com
Country:United States
Release id:44144
The post Tempo Mails Launches Temporary Email Address Platform for Instant Disposable Inbox Access appeared first on King Newswire. This content is provided by a third-party source.. King Newswire makes no warranties or representations in connection with it. King Newswire is a press release distribution agency and does not endorse or verify the claims made in this release. If you have any complaints or copyright concerns related to this article, please contact the company listed in the ‘Media Contact’ section
About Author
Disclaimer: The views, suggestions, and opinions expressed here are the sole responsibility of the experts. No Digi Observer journalist was involved in the writing and production of this article.
-
Press Release6 days ago
TARA BUI AND THE INTERCONTINENTAL SPRINGBOARD STRATEGY: EMPOWERING ASIAN MANUFACTURING GIANTS TO CONQUER THE U.S. MARKET
-
Press Release6 days ago
Smalth Titanium Lava Launches at 88 with Charging Case and Unique Lava Texture
-
Press Release6 days ago
Smart Home Ahead Launches 2026 Survival Guide to End the 168 Billion Dollars Smart Home Frustration
-
Press Release6 days ago
LifeSafe Technologies Highlights Role in Trinity MPF Extinguisher Recognized at Tomorrow’s FM Awards
-
Press Release6 days ago
Digital Smart AI Launches Scalable AI Solutions and GPU Rental Platform for Global Businesses
-
Press Release1 day ago
Leadership That Delivers: How Jeffrey MacBride Is Redefining Project Management Excellence in High-Stakes Industries
-
Press Release1 day ago
El Yam Aluminium Announces General Update on Its Aluminium Pergola Systems and Market Focus
-
Press Release1 day ago
Tempo Mails Launches Temporary Email Address Platform for Instant Disposable Inbox Access



